Investigating Medical Device Failure using Advanced Material and Failure Analyses
by Scott R. Lucas, PHD, PE
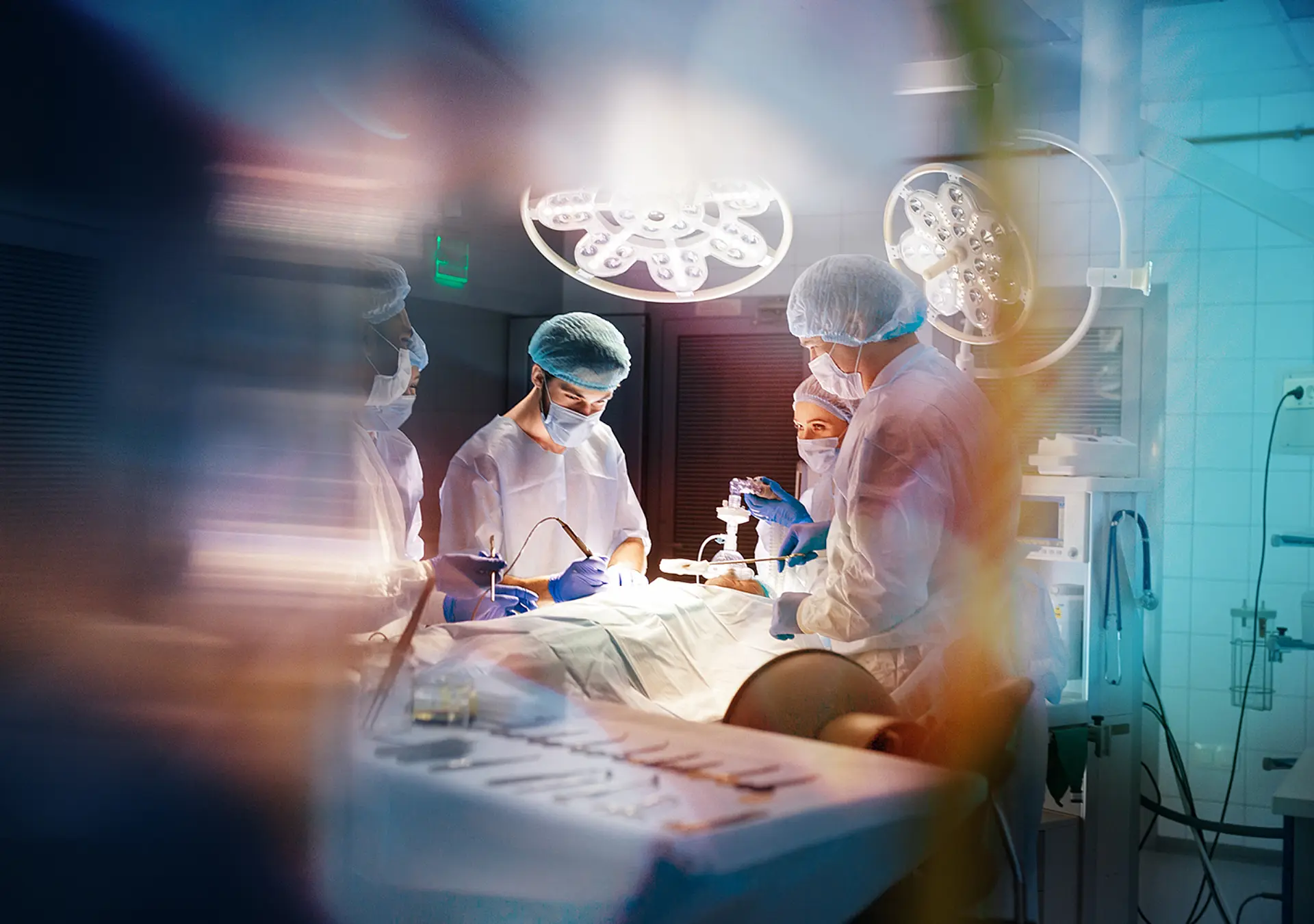
The FDA MAUDE database includes thousands of patient injury reports associated with broken, twisted, perforated, or otherwise damaged medical devices. There are hundreds of articles in the clinical literature on broken surgical instruments alone. Why did these devices fail? Was it a manufacturing or assembly defect? Could it have been user error? Was device integrity affected by cleaning agents? An engineering-based medical device failure analysis can delineate these possibilities. This is especially important in injury forensics to determine causation.
Investigations of medical device failures should be comprehensive and involve a systems-based approach, including cognitive interviews of those involved in the use and maintenance of the device, event simulation, and review of pertinent medical records.
Investigations should also incorporate engineering device failure analyses. Failure analyses include gross and microscopic device inspections as well as advanced techniques, such as:
- Thermal analysis to verify melting temperature, crystallization enthalpy and oxidative induction time.
- Elemental analysis of polymers, metals, ceramics and composites.
- High-resolution metallography and surface morphology to examine failure surfaces.
- Spectroscopy to determine molecular structure of polymers.
Advanced material analysis is also indicated for the identification of unknown materials. Unintended retention of foreign objects (URFO) is the second most reported sentinel event to The Joint Commission. Pieces of guidewire, procedural stents, drains, fragments of broken trocars, metallic shavings from laparoscopic instruments, and many other devices, instruments or fragments can be left behind following a procedure.
In some cases, a foreign body can be identified by context clues (e.g., a sponge left behind from an open surgery). But even in these cases, it may not be clear when the object was left behind (e.g., which surgery resulted in the misadventure). In addition, any contaminant found in the sterile environment on an instrument, in an instrument tray or in the surgical field can be identified using the techniques described herein. Including possible exemplar materials when identifying contaminants is very helpful.
We recommend that healthcare facilities and providers integrate material identification and medical device failure analysis into the regular investigative workflow of medical device forensics. This includes:
- Developing procedures for proper evidence collection and sequestering of broken or damaged medical devices.
- Developing procedures for gross and microscopic inspection of devices.
- Dedicating a light microscope with photogrammetry capability for microscopic inspection.
- Identifying laboratory capability for advanced failure and material analysis.
- Involving a metallurgist, industrial chemist, or appropriate materials scientist to interpret findings from advanced analysis.
- Establishing policies for handling damaged instruments and devices with the manufacturer and patient, which will vary depending on the type of device and whether a patient implant is affected.
- Involving facility risk management and legal counsel in the development of these policies.
Jensen Hughes is proud to support all areas of healthcare in the pursuit of high reliability and zero harm. Learn more about our biomechanics forensic injury investigation services.